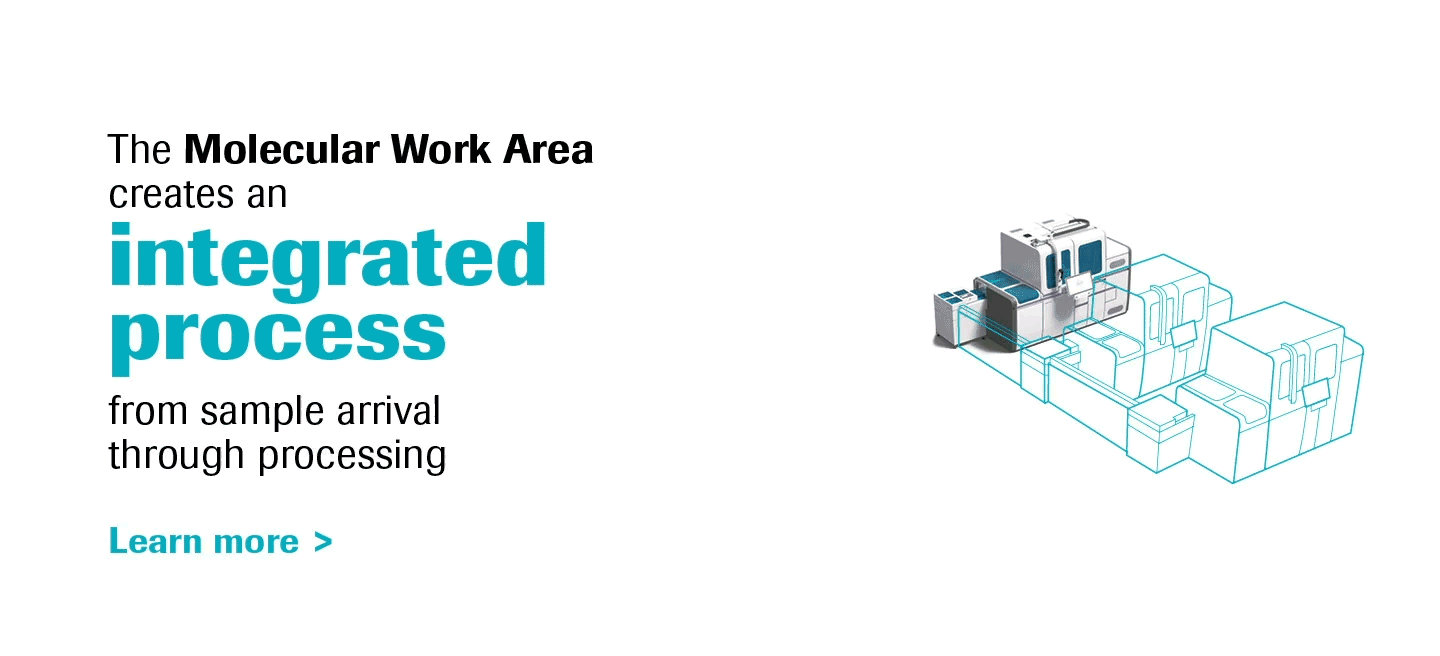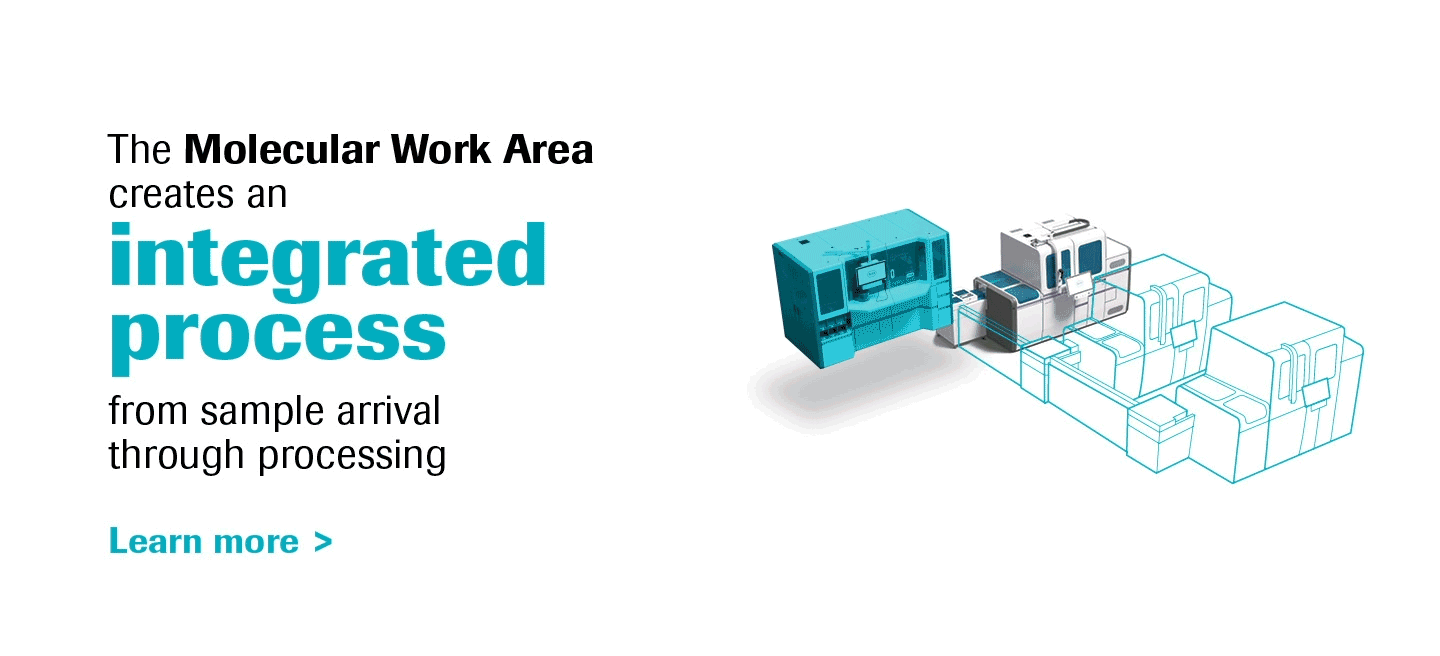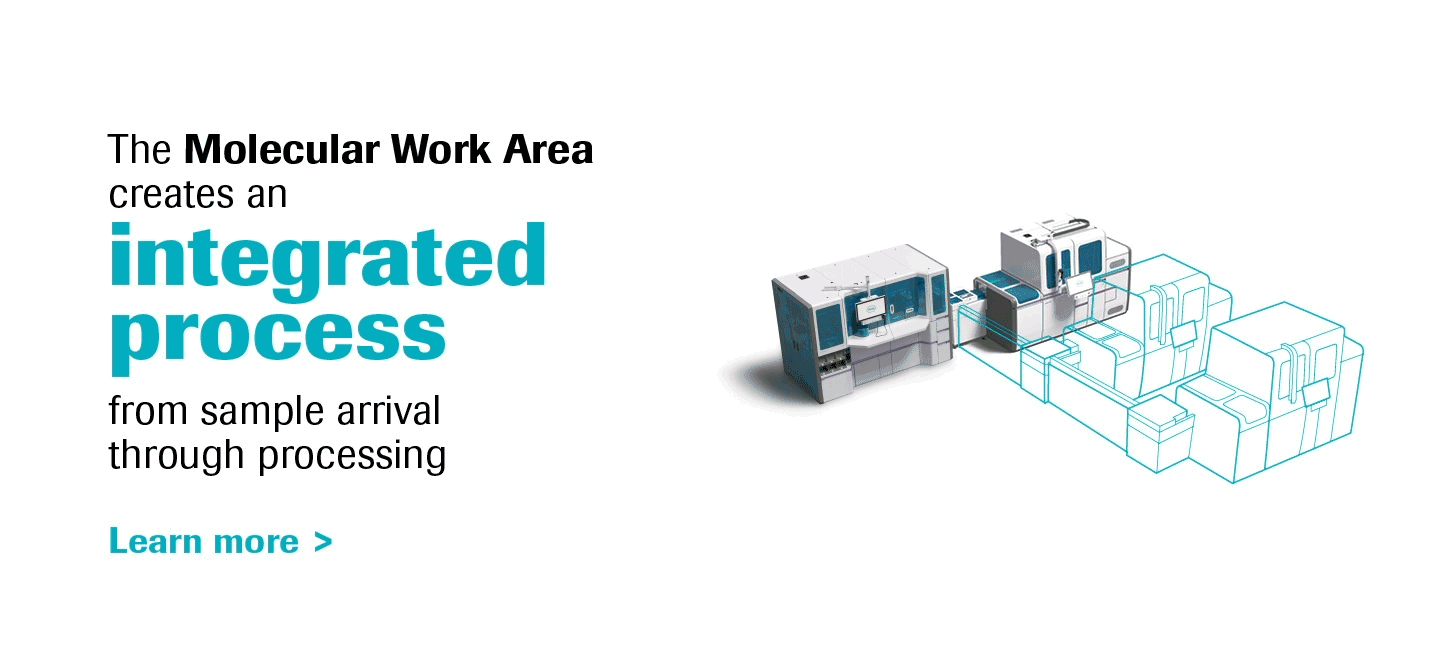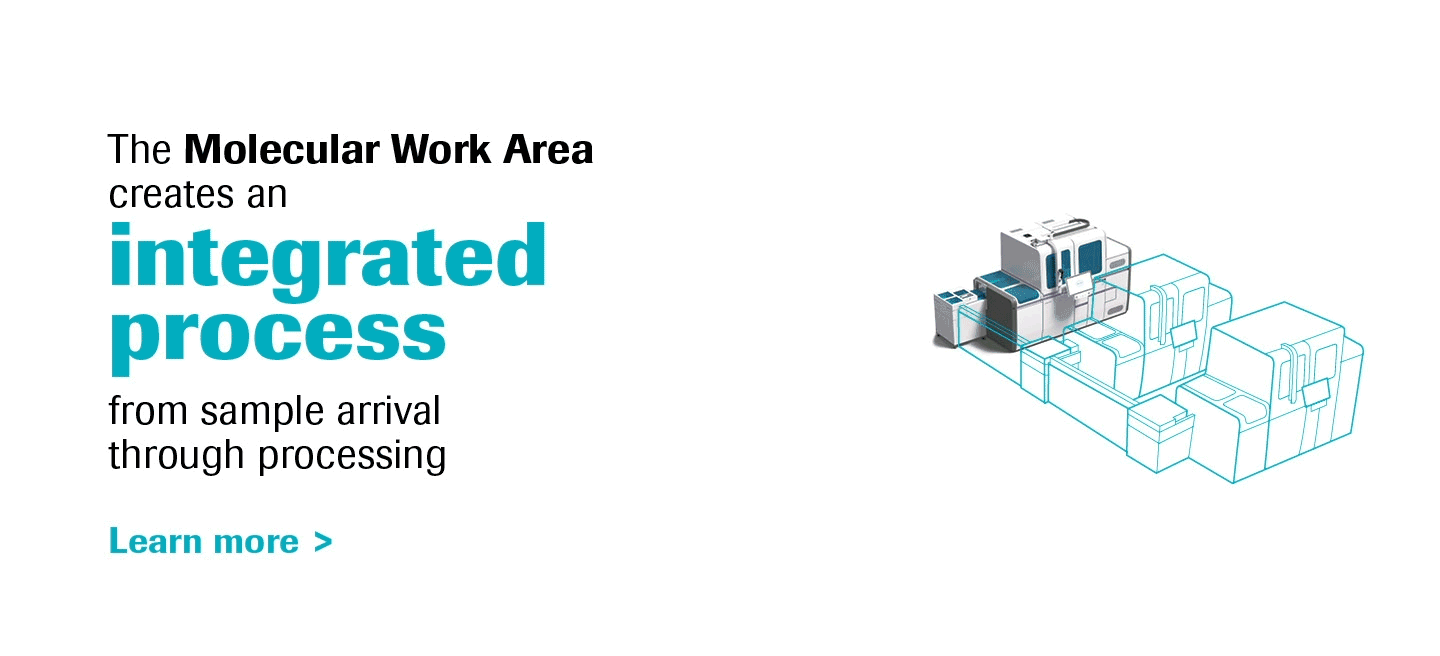
Molecular diagnostics are a key component of laboratory medicine, and PCR is a prime driver of this dynamic.
As the most innovative molecular diagnostics systems on the market, the cobas® 6800/8800 Systems will transform the way molecular labs think and work, truly representing the future of molecular testing.
With increased work-away time labs can create a more meaningful work environment, and the ability to scale efficiently opens a world of possibilities for operational growth.