For localized information and support, would you like to switch to your country-specific website for {0}?
Unleashing the power of the intelligent lab
Transform your core lab
Imagine more connected healthcare settings and agile diagnostic labs, where digital technologies unlock new possibilities for better care.
In shifting a focus on volume to a value-based approach to care, laboratory management and healthcare systems including healthcare delivery settings face many significant shared challenges. These include disconnected data sources and laboratory information systems, declining funding, and a shortage of skilled staff.
navify® digital solutions from Roche Diagnostics help address these challenges by combining connectivity, interoperability, and data privacy/security in an open and secure ecosystem, enabling confident, timely healthcare decisions that can impact patient care, while also helping to drive operational, clinical, and financial value.
"At Roche, we are using our healthcare and technology experience to build open digital ecosystems and solutions with partners to facilitate data access, maintain privacy, and address interoperability. Combined with our diverse, growing digital solution portfolio, we are helping labs and hospitals unlock new opportunities for their clinicians and patients to benefit from personalized care approaches."
- Matt Manley, Vice President, Digital Healthcare Solutions at Roche Diagnostics.
Improve lab operational efficiency with digital solutions
Staff shortage, administrative burdens, data overload, and increasing cost of care are some of the challenges that healthcare is facing today.
By connecting data and insights, our navify digital solutions for core lab can help to:1
- Deliver faster and more accurate diagnoses by leveraging medical algorithms
- Reduce operational burdens and maximize efficiencies by improving workflows
- Support clinical decisions across the care continuum to support more personalized care.
Connected insights for better patient care
We strive for a more connected and agile healthcare system, where digital technologies unlock new possibilities for better care. We are committed to differentiating your lab from the competition and enabling you to impact patient care with our medical insights solutions.
What inspires our work:
- Connecting patient data to enable the transition to patient-centric care
- Bringing healthcare thinking to technology to enable value-based care
- Co-creating interoperable solutions to accelerate access to innovation.
Featured products
Benefits of navify digital solutions for core lab from Roche
Manage a secure and better-connected lab
Our digital products work together to solve some of your challenges.
- Increase productivity by optimizing workflows
- Use your data to identify bottlenecks and proactively mitigate issues
- Monitor sample tracking
- Continuously adjust for optimization
- Optimize the use of reagents and quality control (QC) materials
- Identify reagents with low usage efficiency
- Set up the instrument throughput to quickly react to occurring bottlenecks and identify periods of low instrument usage
- Prove improved reagent usage and demonstrate a positive impact on turnaround time (TAT)
- Improve reaction times on critical results
- Leverage data that can help define normal and critical ranges of test results
- Implement the threshold for the critical range and automate the validation of results
- Flag any results that have reached the threshold
- Review if the reaction time is within the target range
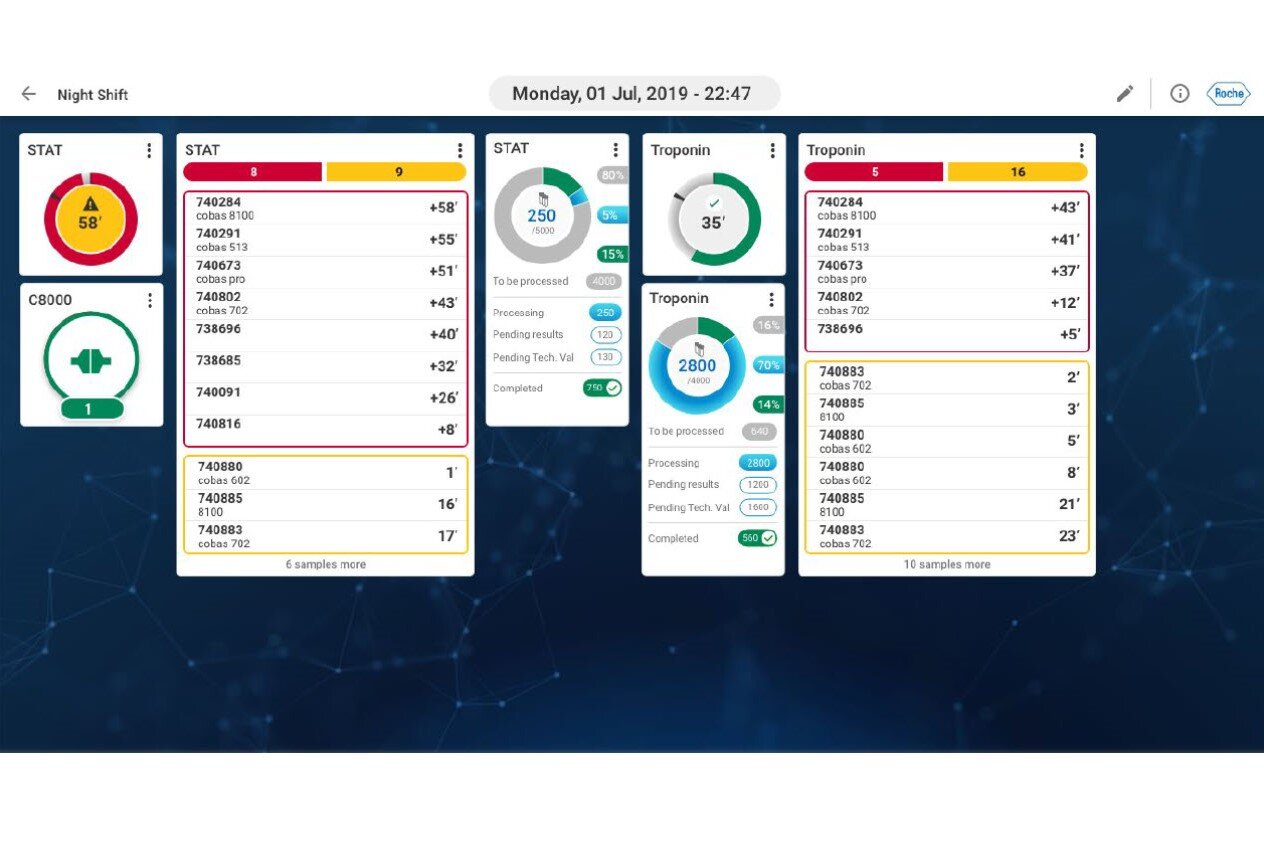
Enhance efficiency, testing capacity, and operational transparency
- Drive operational effectiveness: Simplify laboratory processes and workflows with enhanced sample, result, and data management, and extended connectivity
- Take charge of lab monitoring: Real-time reporting and remote control through the command cockpit
- Unlock the potential of lab data: Instrument utilization analytics, retrospective and c-suite reporting, and insights for enhanced outputs
Secure and scalable solutions that can be personalized to your needs
- Robust cybersecurity: Implement multiple layers of security controls for continuous monitoring
- Data privacy and security: Benefit from Roche's secure-by-design cloud architecture to help address data privacy and localization commitments
- Compliance support: Leverage Roche’s global expertise and processes to meet government regulatory requirements
- Reliability and uptime: Experience robust resiliency and disaster recovery capabilities to help maximize system uptime

Transforming lab management with navify® digital solutions
Burjeel Holdings selected navify® Lab Operations to digitalize laboratory operations across their 13 hospitals. Learn how it reduced key tasks and turnaround times, helping improve service and satisfaction for physicians and patients.

NAVIFY is a trademark of Roche.
References:
- Roche Diagnostics Ltd. Data on file. MC--10689