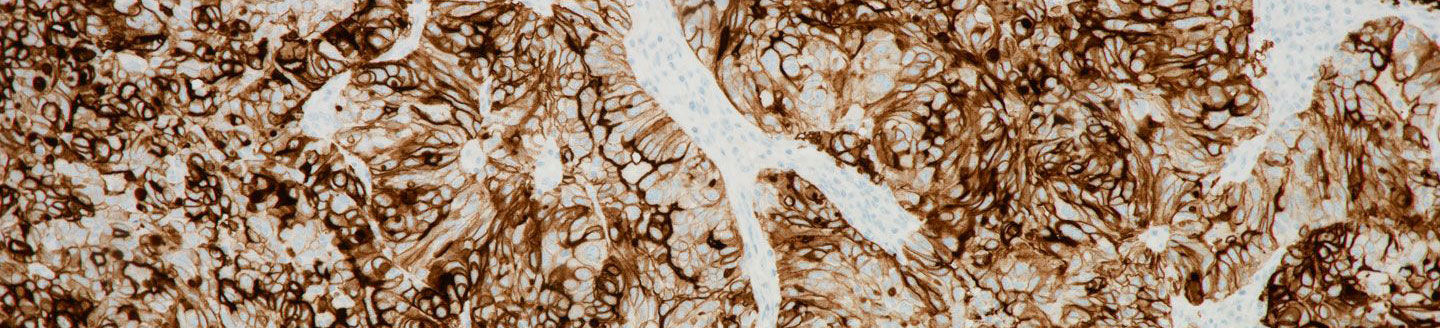
- The VENTANA FOLR1 (FOLR1-2.1) RxDx Assay detects the folate receptor 1 protein (FOLR1 or FRɑ), which is over-expressed in most ovarian cancers.
- The test identifies ovarian cancer patients eligible for targeted treatment with ELAHERE.
- This certification follows the unprecedented decision by regulators to grant an early exemption approval for the test in Germany and Austria earlier this year.
Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that it has received CE Mark for its VENTANA® FOLR1 (FOLR1-2.1) RxDx Assay. This is the first immunohistochemistry (IHC) companion diagnostic test to be made widely available in Europe to aid in identifying epithelial ovarian cancer (EOC) patients who may be eligible for targeted treatment with ELAHERE® (mirvetuximab soravtansine). ELAHERE is a first-in-class antibody-drug conjugate (ADC) therapy developed by AbbVie for the treatment of FRɑ-positive platinum-resistant ovarian cancer.
This certification follows the news earlier this year that the VENTANA FOLR1 (FOLR1-2.1) test had received pre-authorisation in Germany and Austria. This exceptional decision allowed clinicians and patients in these countries to access the test ahead of the CE Mark certification.
“This certification will allow us to extend the reach of our innovative diagnostic solutions,” said Jill German, Head of Pathology Lab at Roche Diagnostics. “The early exemption approval in Germany and Austria highlighted the urgent need for this test. Now, clinicians across Europe can access a critical tool to quickly identify ovarian cancer patients who may be eligible for targeted therapy. By enabling more precise and personalised treatment decisions, we hope this may help improve outcomes for the many women in Europe facing this devastating disease.”
Folate receptor 1 protein (FOLR1), also known as folate receptor alpha (FRɑ), is expressed at some level in approximately 90 percent of ovarian carcinomas and serves as a predictive biomarker for FOLR1-targeted therapy for EOC patients.1,2 The VENTANA FOLR-1 (FOLR1-2.1) test informs clinicians about the likelihood of potential patient benefit from FOLR1 therapy,3,4 advancing Roche’s commitment to personalised healthcare through innovative solutions that help fit the treatment to the individual.
Ovarian cancer is the eighth overall cause for cancer death in women worldwide, representing 4.7% of all cancer deaths in women. It is also one of the deadliest gynaecological cancers worldwide. In 2022, 46,232 women in Europe and 209,596 women worldwide died from ovarian cancer.5
The launch of Roche’s first IHC companion test for ovarian cancer in CE countries highlights the company’s commitment, as the world’s leading provider of in vitro diagnostics, to continued innovation and evolution of its products in order to advance personalised healthcare and deliver novel, high medical value solutions that improve patients’ lives.