Grâce à son automatisation et sa connectivité, du traitement des échantillons à l'interprétation des résultats, le système cobas® 8800 vous permet d’être équipé pour l’avenir.
cobas® 8800 system

La durabilité à long terme est là1
Quelque soit la discipline, libérez votre capacité à automatiser et à consolider jusqu'à 90% des tests de routine en un seul flux de travail. Avec 4 heures d'autonomie de travail et seulement 3 interventions utilisateur par journée de travail, le personnel du laboratoire est libre de se concentrer sur des tâches à plus grande valeur ajoutée.

* En développement
‡ Auto-prélèvement disponible
§ cobas omni Utility Channel CE-IVD
# cobas® Plasma Separation Card
Depuis 2014, plus de 1500 systèmes cobas® 6800 et cobas® 8800 ont été installés sur les cinq continents, aidant les laboratoires à automatiser, consolider, intégrer et standardiser les tests moléculaires comme jamais auparavant.
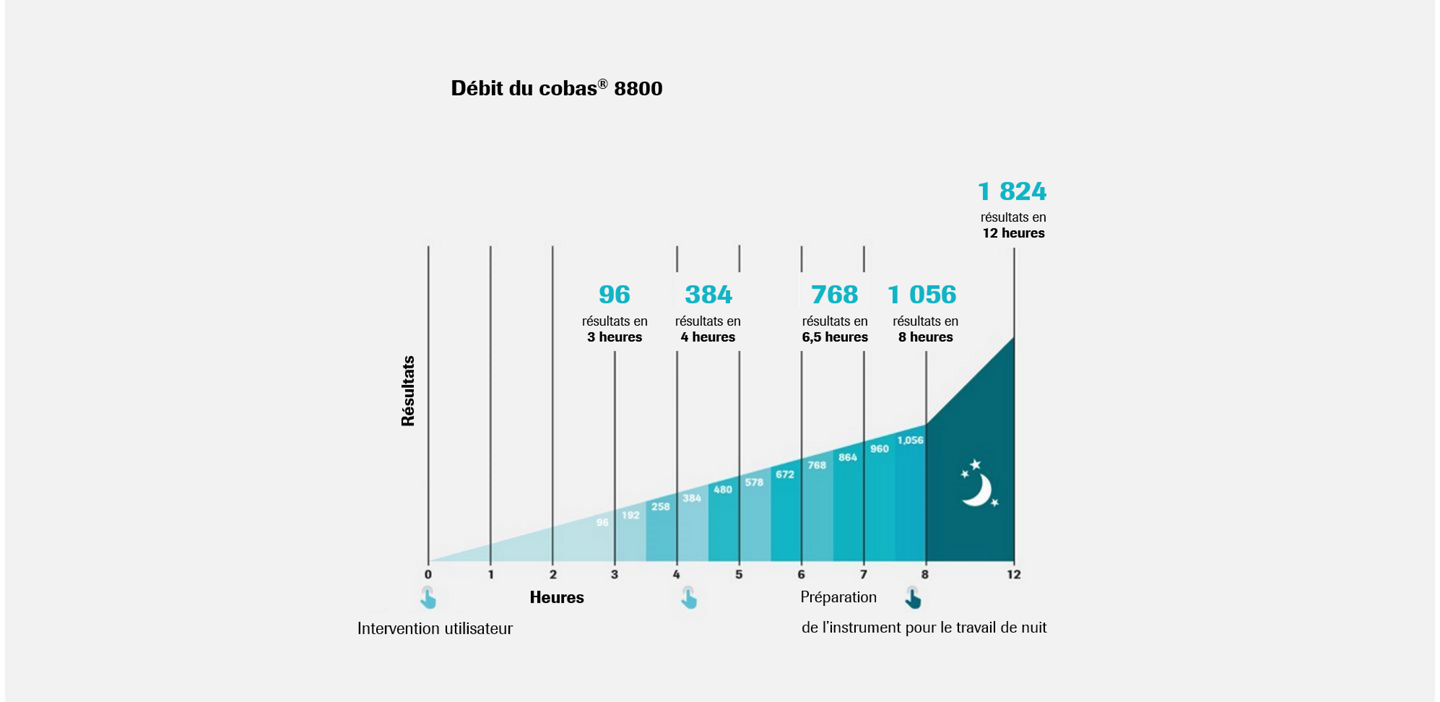
Exécution rapide des tests quotidiens, avec des résultats fiables et reproductibles.1
- Temps de traitement rapide, jusqu’à 96 tests en environ 3 heures
- Jusqu'à 1056 échantillons pour 8 heures de travail
- Manipulation minimale, seulement trois interventions techniques
- Les réactifs prêts à l'emploi ne nécessitent ni décongélation, ni mélange, ni manipulation tandis que l’identification par radiofréquence (RFID) et les codes-barres garantissent une traçabilité complète.
- Le système automatisé de stockage et de refroidissement à bord permet un accès direct aux consommables et aux réactifs dans le système
- L’interface unidirectionnelle et bidirectionnelle du système de gestion du laboratoire (SGL) simplifie le traitement des commandes et des résultats
- Possibilité de connecter jusqu'à 6 instruments cobas p 680 pour le poolage automatisé, l'archivage des échantillons et la traçabilité du déroulement des opérations

* Peuvent varier en fonction des demandes de flux de travail
- Possibilité de combiner les tests : jusqu'à 3 tests par plaque PCR
- Optimisation de l’utilisation des échantillons grâce à la possibilité d’effectuer jusqu’à trois tests différents sur un même échantillon
- Simplification des flux de travail grâce à des processus, des consommables et des réactifs prêts à l'emploi
- Traitement rapide et facile des échantillons urgents grâce à une voie prioritaire dédiée
Vous souhaitez être recontacté ?
Remplissez le formulaire ci-contre pour être recontacté par un expert de Roche Diagnostics France. Il vous fournira des informations personnalisées sur le cobas® 6800 et étudiera avec vous la façon dont il peut s'intégrer dans votre environnement de travail, et pourra vous faire bénéficier d'une démonstration pratique de son utilisation.
Nous nous engageons à vous recontacter dans les plus brefs délais pour répondre à toutes vos questions concernant le cobas® 6800 .
Les données collectées dans le cadre de ce formulaire sont traitées par Roche Diagnostics France, responsable de traitement et destinataire des données, pour :
- répondre à la demande que vous nous adressez (inscription, accès au replay, renseignement, contact...). Les données sont traitées sur la base de notre intérêt légitime et sont conservées dans notre outil de gestion de la relation client pour être utilisées dans le cadre de la relation commerciale ou à des fins de prospection commerciale physique.
- si vous ne vous y opposez pas, vous adresser des communications marketing ciblées, en lien avec votre profession, et ce, sur la base de notre intérêt légitime. Vous pouvez vous opposer à ce traitement à tout moment, en cliquant sur le lien de désinscription présent dans les e-mails qui vous sont adressés.
Les données sont conservées pendant 3 ans à compter de la fin de la relation commerciale ou 3 ans à compter du dernier contact.
Conformément à la législation en vigueur et notamment au Règlement Général sur la Protection des Données Personnelles du 27 avril 2016, vous disposez notamment d’un droit d’accès, de rectification, d’effacement, de portabilité de vos données personnelles. Vous pouvez limiter ou vous opposer au traitement de celles-ci pour l’avenir en écrivant à [email protected].
Si vous estimez que vos droits individuels ne sont pas respectés, vous pouvez faire une réclamation à la CNIL.
Pour en savoir plus, consultez notre Politique en matière de respect de la vie privée.
Si vous ne souhaitez plus recevoir nos communications marketing, merci de cliquer sur "désinscription".
Référence :
- Guide utilisateur cobas 6800/8800, v5.1, 05/2022
Les cobas® 6800/8800 Systems permettent l'automatisation et l'intégration du flux de travail pour la réalisation de tests d'acide nucléique (ou NAT pour Nucleic Acid Testing) basés sur la réaction en chaîne par polymérase (ou PCR pour Polymerase Chain Reaction).
Dispositif médical de diagnostic in vitro. Classe A
Mandataire : Roche Diagnostics GmbH (Allemagne) – Distributeur : Roche Diagnostics France
Lire attentivement les instructions figurant dans le manuel d’utilisation.
Mis à jour : 01/2023 - MC-FR-01734



